

Raman spectroscopy is a non-destructive vibrational spectroscopy technique to identify a fingerprint of a material based on vibrational frequencies of the various atomic bonds in the material. Raman spectroscopy is a complementary technique to IR absorption spectroscopy, but provides a plethora of information about the samples other than identification. Raman spectroscopy can be used with solid, liquid and gaseous samples. Raman spectroscopy facility includes three lasers (532nm, 633nm and 785 nm) and a heating/cooling stage for samples. The spectrometer range is from 50 to 4000 cm-1 (Raman shift) and max resolution of 0.5 cm-1. Raman spectroscopy can be utilized for identification of materials (chemical bonds), phase (symmetry), and calculation of stress (change in bond lengths) and probing of electronic structure of nanomaterial's |
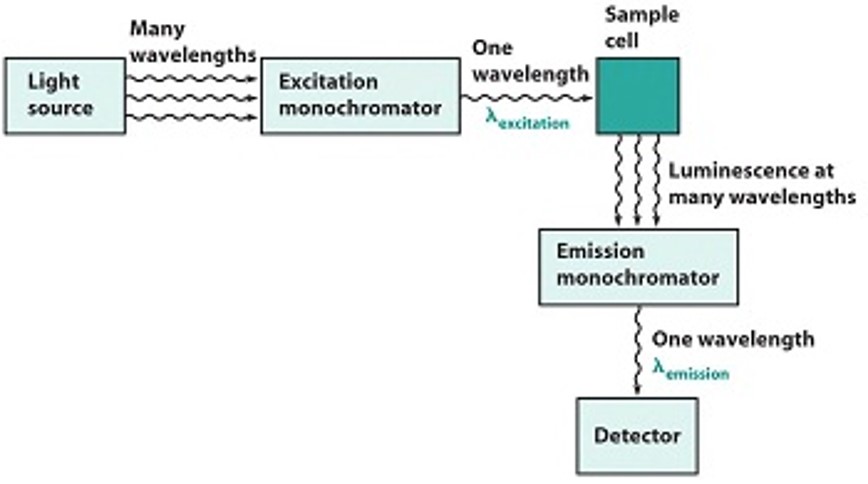
Photoluminescence spectroscopy is a contactless, nondestructive method of probing the electronic structure of materials. Light is directed onto a sample, where it is absorbed and imparts excess energy into the material in a process called photo-excitation. One way this excess energy can be dissipated by the sample is through the emission of light, or luminescence. In the case of photo-excitation, this luminescence is called photoluminescence. |