Professor Singh's research work falls in the area of synthetic organic chemistry, more specifically, asymmetric synthesis. Prof. Singh has accomplished total synthesis of several bioactive natural products and medicinally important compounds and had developed a number of novel asymmetric methodologies for the synthesis of several optically pure and therapeutically as well as pharmacologically useful chiral building blocks of immense synthetic importance.
His initial research in the area of asymmetric synthesis particularly on enantioselective deprotonation of epoxides and allylic oxidation of olefins, received high appreciation from world all over.
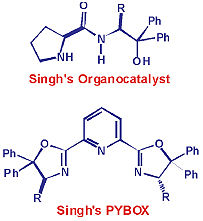
Currently, Prof. Singh is working in the area of Asymmetric Catalysis, which is one of the most important and cutting-edge area of research in Synthetic Chemistry. Towards the metal-catalyzed enantioselective transformations, his group has successfully applied iPr-Pybox-diPh ligands in enantioselective allylic oxidation of olefins and enantiosele-ctivepropargylation reactions.
In 2008, the group explored bidentate chelating substrate 2-enoylpyridine N-oxide as a new template in asymmetric Michael reactions. Using this template they carried out enantioselective Michael reactions of indoles, pyrroles, dialkylmalonates, 4-hydroxy coumarins, and 1,3-dicarbonyls and Mukaiyama-Michael reaction as well using sillylenolethers thus expanding the scope of iPr-Pybox-diPh ligands.
In 2006, Singh and his research group designed a new organocatalyst, popularly known as Singh's catalyst for asymmetric Aldol reaction. This is one of the best catalysts known for enantioselective aldol reactions till date.
Another area where Singh's group contributed significantly is the enan-tioselectiveorganocatalytic reactions using H-bonding catalysts via dual activation mode.
-
Asymmetric Alkynylation/Lactamization Cadcade: An Expeditious Entry to Enantiomerically Enriched Isoindolinones Angew. Chem. Int. Ed., 53, 10737 (2014).
-
Highly Enantioselective Conjugate Addition of Malonitrile to 2-Enoylpyridines with Bifunctional Organocatalyst, Org. Lett., 14, 4322 (2012).
-
Enantioselective Michael Addition of Malonates to 2-Enoylpyridine N-oxides Catalyzed by Chiral Bisoxazoline-Zn(II) Complex, Org. Lett., 13, 5812 (2011).
-
Enantioselective Enolate Protonation in Sulfa-Michael Addition to a-Substituted N-acryloyloxazolidin-2-ones with Bifunctional Organocatalyst, Org. Lett., 13, 6520 (2011).
-
Enantioselective Friedel-Crafts Alkylation of Pyrroles Catalyzed by Pybox-Diph-Zn(II) Complexes, Org. Lett., 12, 80 (2010).
-
Highly Enantioselective Organocatalytic Sulfa-Michael Addition to α, β-Unsaturated Ketones, J. Org. Chem., 75, 2089 (2010).
-
Highly Efficient Small Organic Molecules for Enantioselective Direct Aldol Reaction in both Organic and Aqueous Medium: Application in Synthesis, J. Org. Chem., 74, 4289 (2009).
-
Organocatalytic Reactions in Water, Chem. Commun., 6687 (2009).
-
Highly Enantioselective Friedel-Crafts Reaction of Indoles with 2-Enoylpyridine 1-Oxides Catalyzed by Chiral Pyridine 2,6-Bis (5’,5’-diphenyloxazoline)-Cu(II) Complexes, Org. Lett., 10, 4121 (2008).
-
Founder Director, Indian Institute of Science Education and Research, Bhopal, 2008.
-
Harvard University (Advisor: Prof. E. J. Corey), USA, 1987-1990
-
Member, Scientific Advisory Council to the Prime Minister (SAC to PM)
-
Founder Director, Indian Institute of Science Education and Research, Bhopal, 2008.
-
Padma Shri, 2014.
-
Distinguished Alumnus Award, BHU, 2012
-
Fellow, Indian National Science Academy, 2011
-
Goyal Prize, 2011
-
Shanti Swarup Bhatnagar Prize, 2004
-
Swarnajayanti Fellowship, 1998
Office
CL 205A ,
Department of Chemistry
IIT Kanpur,
Kanpur 208016
Office Phone: 0512-259-7291 (O)
Email: vinodks[AT]iitk.ac.in
|
|
|












